Beyond the heptad repeat, a range of other periodicities is accessible to coiled coils, which is only restrained by the periodicity of the unperturbed α-helix ( Gruber and Lupas, 2003). The seven positions in a heptad are labeled a – g, where positions a and d are oriented towards the core of the bundle and are thus mostly hydrophobic. In their most prevalent form they follow a heptad sequence repeat pattern. They consist of at least two α-helices, wound into superhelical bundles and held together by a mostly hydrophobic core. The structure of coiled coils is understood at a level unrivaled by any other fold. They were the first protein fold described in atomic detail ( Crick, 1953b) and are also the only one whose backbone structure can be computed with parametric equations ( Crick, 1953a), placing them at the forefront of protein design efforts ( Huang et al., 2014 Joh et al., 2014 Thomson et al., 2014 Woolfson, 2005). Α-Helical coiled coils are ubiquitous protein domains, found in a wide range of structural and functional contexts ( Lupas, 1996). Hartmann et al.’s findings suggest that the alternating α-helix and β-strand structures may help to make the proteins stronger and enable to carry out more versatile roles in cells. named a β-layer.įurther experiments showed that inserting the same numbers of amino acids into the heptad repeats of other coiled coil proteins also resulted in the formation of β-layers. The three short β-strands then associate into a triangular structure that Hartmann et al. The experiments show that inserting these numbers of amino acids caused so much strain that the three α-helices making up the coiled coil break apart and refold into a completely different type of structure called a β-strand. These numbers of amino acids have been predicted to cause the greatest strain on the coiled coil structure. investigated what would happen when two or six amino acids were inserted into the heptad repeats of a coiled coil within a protein from bacteria. three or four at a time) has been studied, the effect of inserting other amounts of amino acids was not clear. Although the impact of some insertions (e.g. 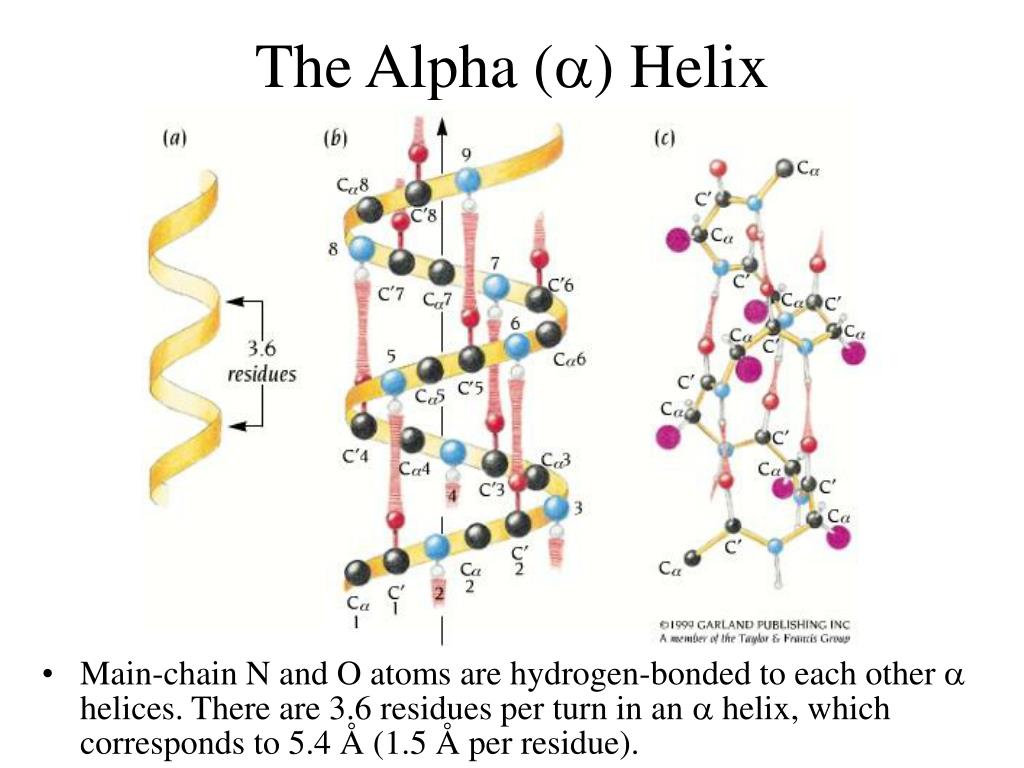
This may be due to insertions of small numbers of amino acids. Many naturally occurring coiled coils have regions where the overall α-helical structure is retained, even though there are small sections where the number of amino acids in a turn is disrupted. However, it cannot increase too far beyond the number of amino acids in each turn of a normal α-helix because there is a limit to the amount of coiling that the helices can tolerate. When amino acids are added into or deleted from the heptad repeats, the number of amino acids in the turns of a coiled coil changes. The most common pattern in a coiled coil has 7 amino acids over two turns, which is known as a heptad repeat. Each complete turn of an α-helix contains a set number of amino acids, but the number of amino acids in the turns of a coiled coil can vary. 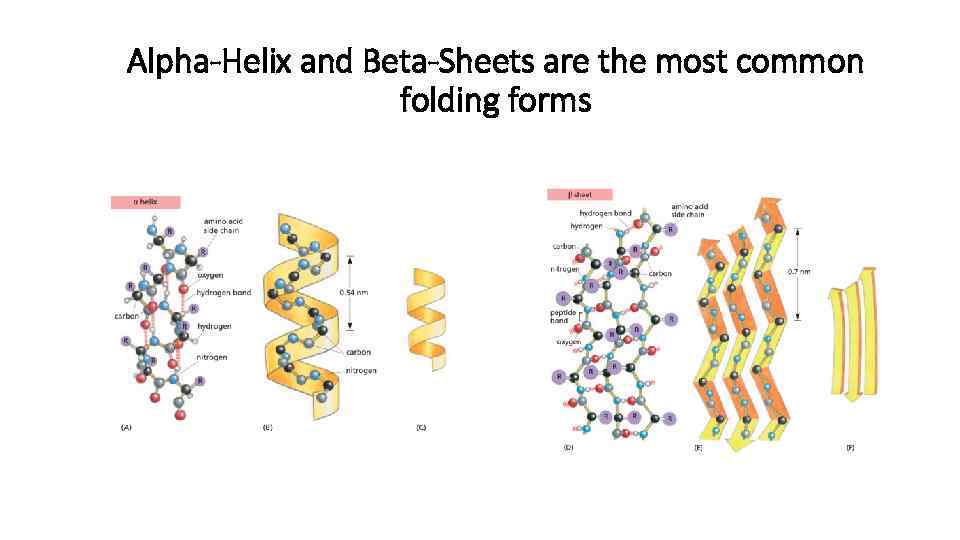
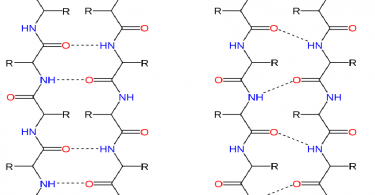
Two or more α-helices may be wound around each other to form a bundle called a coiled coil, which is found in many proteins. Groups of amino acids within the protein can then fold into three-dimensional shapes, one of the most common being a helical structure known as an α-helix. Proteins are made up of building blocks called amino acids.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
